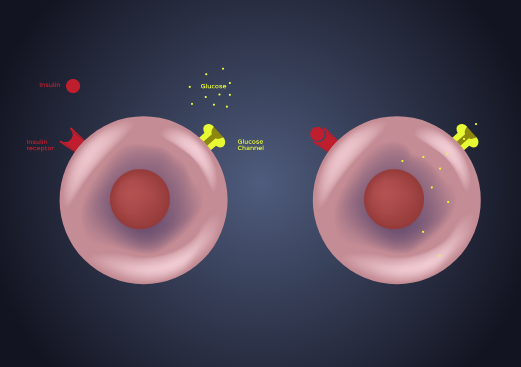
Introduction
You are being asked to participate in a research study. Before you decide to participate in this study, it is important that you understand why the research is being done and what it will involve. This form is designed to give you the information about the study so you can decide whether to be in the study or not. Please take the time to read the following information carefully. You should ask the researcher if there is anything that is not clear or if you need more information.
Study Participation
If you agree to participate in this study, you will be asked to:
- Attend screening visit to determine eligibility
- Attend 6–8 clinic visits over approximately 12 weeks
- Provide blood samples at designated visits
- Complete questionnaires about your symptoms and quality of life
- Take study medication or placebo as directed
- Report any side effects or health changes to the study team